PhD
Distinguished Professor, Microbiology and Immunology, Biochemistry and Biophysics
UNC-Chapel Hill
Virology
Area of Interest
Our laboratory continues to focus on the use of electron microscopy (EM) to elucidate broad mechanisms of DNA protein interactions as they impinge on cancer and oncogenesis. Three areas central to cancer merit particular review: basic studies of DNA replication, studies of DNA repair and the involvement of p53 as a sensor of DNA damage and studies of the structure of human telomeres.
a) Studies of DNA replication in model systems and in the oncogenic human viruses HSV and HPV. Our work on DNA replication has resulted in two publications with Charles Richardson and his laboratory that have provided firm proof of the trombone model of DNA replication, a model that has been seeking such direct proof since it was presented by Bruce Alberts 25 years ago. In a paper in J Biol Chem we demonstrated that the T7 replication system is able to replicate DNA in a coordinated manner and that trombone loops involving the lagging strand can be directly seen by EM. In a paper that just appeared in Mol Cell we show that a tiny DNA template of 70 base pairs is also replicated by the trombone mechanism, and that the tiny circle rotates within the protein machinery at 240 RPM.
Using a combination of EM and biochemical approaches we have collaborated with Robert Lehman at Stanford to study the mechanism by which two HSV proteins, UL9 and ICP8 activate the origin of Herpes Simplex virus. Previously there had been no direct evidence that the UL9 origin binding protein could unwind the HSV origin. In a paper in EMBO J, we demonstrated that UL9 can unwind the HSV origin and in an ATP dependent manner will generate large loops of unwound DNA. In work nearing publication we have extended this finding to include studies of how the ICP8 protein facilitates the unwinding step. This is a very important area of HSV research since it has the potential to identify new drug target steps. In work with Drs. Tom Broker and Louise Chow, we have initiated studies (submitted for publication) on how the human papilloma virus E1 protein activates the origin of HPV and how this process is greatly augmented by two human heat shock proteins HSP40 and HSP70.
b) Studies of DNA mismatch repair and the role of p53 protein. Our laboratory has had a longstanding interest in the mechanism by which the mismatch repair proteins identify damage in DNA. This goes back 12 years in studies of the E coli mutS protein with Paul Modrich at Duke University and our own studies of how the recA protein will recognize certain damage in DNA. A key paper was published with the Modrich laboratory in 1997 (cover of EMBO J) demonstrating that the mutHSL system identifies damage in DNA by a mechanism that involves the mutS induced formation of a loop in the DNA. The loop grows in size beginning at a mismatch in an ATP dependent manner until a signal (hemi methylated GATC) is found. This then signals the subsequent repair steps.
Awards and Honors
- Progress in Photography Award, Photographic Society of America, 2020
News and Stories
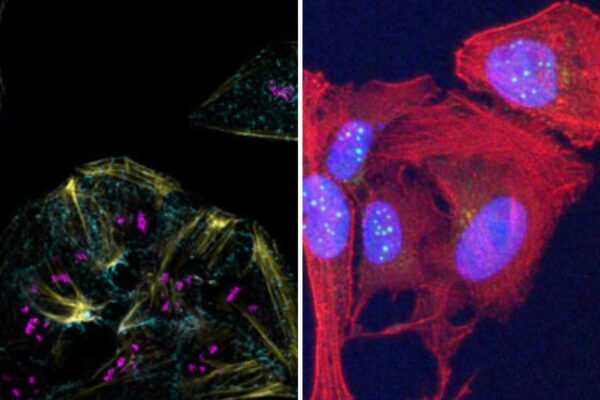
Dittmer, Griffith labs win Art in Science Competition
During University Research Week, the labs of Dirk Dittmer, PhD, and Jack Griffith, PhD, won the Art in Science Competition held by the UNC School of Medicine Office of Research.
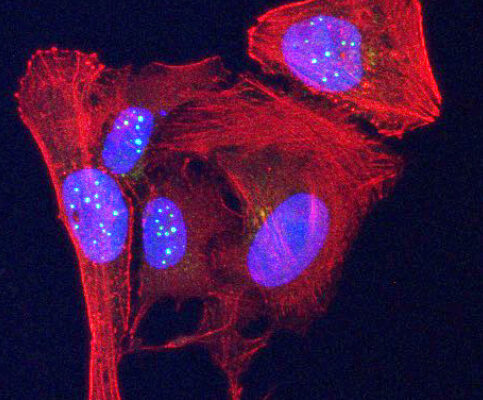
Scientists make stunning discovery, find new protein activity in telomeres
UNC Lineberger researchers made the stunning discovery that telomeres contain genetic information to produce two small proteins, one of which they found is elevated in some human cancer cells.

Enigmatic protein sculpts DNA to repair harmful damage
Jack Griffith, PhD, co-authored a paper showing the role the XPG protein plays in DNA repair in healthy human cells, and how mutations can translate into diseases and cancer.

Griffith wins Photographic Society of America’s Progress Award
Jack Griffith, PhD, has received the Progress in Photography Award from the Photographic Society of America for his work using photo-microscopy with the electron microscope.

